Cancer risk in heart or lung transplant recipients: a meta-analysis
Fan Ge1, Runchen Wang2, Zhenyu Huo2, Yaokai Wen2, Hengrui Liang4, Yu Jiang2, Zixuan Su2, Wenhua Liang3, Jianxing He4.
11st Clinical School, Guangzhou Medical University, Guangzhou, People's Republic of China; 2Nanshan School, Guangzhou Medical University, Guangzhou, People's Republic of China; 3Department of Thoracic Oncology, The 1st Affiliated Hosipital of Guangzhou Medical University, Guangzhou, People's Republic of China; 4Department of Thoracic Surgery, The 1st Affiliated Hosipital of Guangzhou Medical University, Guangzhou, People's Republic of China
Background: Patients underwent heart or lung transplantation are considered to be associated with an increased risk of developing cancers. However, the specific cancer risk at different sites among heart or lung transplantation recipients remain unclear. Tumor mutational burden (TMB) is a biomarker, which is a measure of immunogenicity of certain cancer.
Methods: PubMed, Web of Science, EMBASE, Medline, Cochrane Library were searched. Random-effects meta-analyses were performed to calculate standardized incidence ratios (SIRs) for heart or lung transplant recipients versus the general population, stratified by types of transplantation. Weighted linear regression (WLR) was performed to analyze the associations between tumors’ standardized incidence ratios (SIRs) and corresponding tumor mutational burden (TMB).
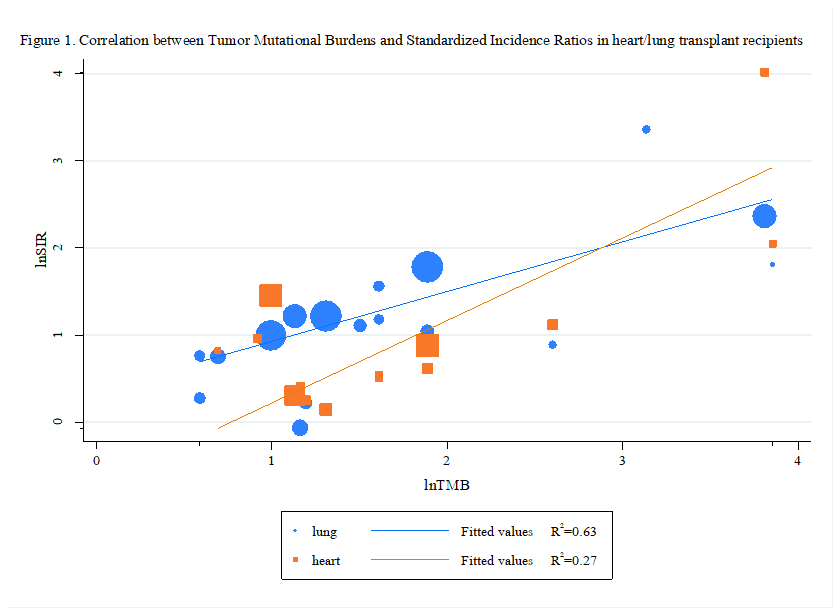
Results: Based on data from 117,045 transplant recipients (51,173 heart transplant recipients and 65,872 lung transplant recipients) with a total follow-up of 578,014.8 person-years (heart transplantation: 259,913.2 person-years; lung transplantation: 318,101.6 person-years), we identified that heart or lung transplant recipients display a different risk in different cancers. Heart transplant recipients were found to display a higher risk in all cancers [SIR: 3.13; 95% Confidence Interval (CI): 2.38-4.13; P<0.001], squamous cell carcinoma, lip cancer, non-melanoma skin cancer, non-Hodgkin's lymphoma, basal cell cancer, kidney cancer, compared with the general population. Lung transplant recipients were found to display a higher risk in all cancers [SIR: 4.28; 95% CI: 2.98-6.60; P < 0.001], non-Hodgkin's lymphoma, lip cancer, skin cancer, non-melanoma skin cancer, squamous cell carcinoma, lung cancer, head and neck cancer, colorectal cancer, liver cancer, cervical cancer and a lower risk in breast cancer, compared with the general population (Table 1). 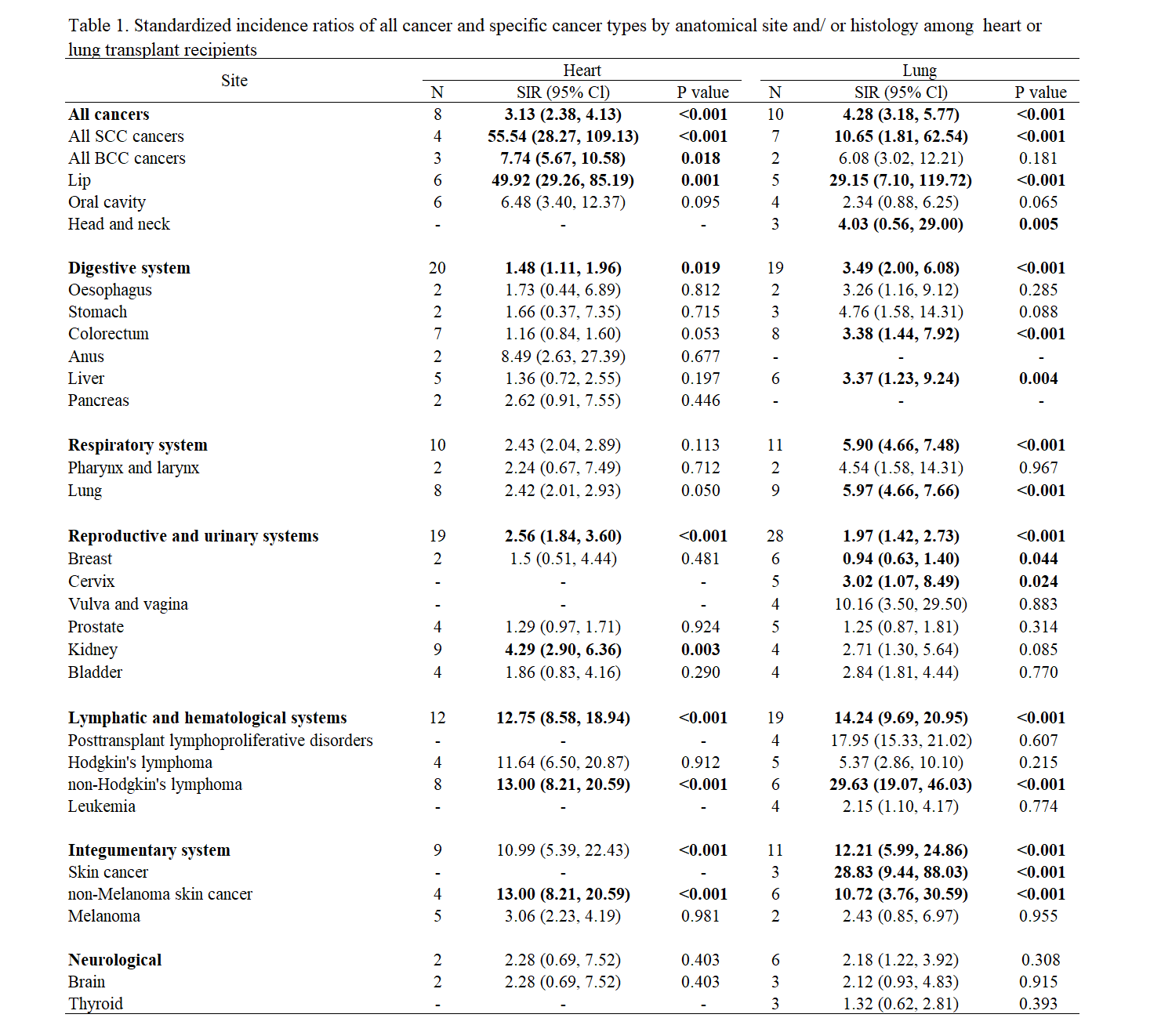 The correlation coefficient of 0.54 [P = 0.047] and 0.79 [P < 0.001] in heart and lung transplant recipients, respectively, suggested that 27% and 63% of the differences in the standardized incidence ratios across cancer types may be explained by the tumor mutational burden (Figure1).
The correlation coefficient of 0.54 [P = 0.047] and 0.79 [P < 0.001] in heart and lung transplant recipients, respectively, suggested that 27% and 63% of the differences in the standardized incidence ratios across cancer types may be explained by the tumor mutational burden (Figure1).
[1] 1. Chambers DC, Cherikh WS, Goldfarb SB, et al.: The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-fifth adult lung and heart-lung transplant report-2018; Focus theme: Multiorgan
[2] 2. Chambers DC, Cherikh WS, Harhay MO, et al.: The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-sixth adult lung and heart-lung transplantation Report-2019; Focus theme: Donor and recipient size match. J Heart Lung Transplant 2019;38:1042-55.
[3] 3. ISHLT Adult Heart Transplantation Statistics. 2018. [cited 2020 Jan 20]. Available from: https://ishltregistries.org/registries/slides.asp.
[4] 4. Aberg F, Pukkala E, Höckerstedt K, Sankila R, Isoniemi H: Risk of malignant neoplasms after liver transplantation: a population-based study. Liver Transpl 2008;14:1428-36.
[5] 5. Adami J, Gäbel H, Lindelöf B, et al.: Cancer risk following organ transplantation: a nationwide cohort study in Sweden. Br J Cancer 2003;89:1221-7.
[6] 6. Engels EA, Pfeiffer RM, Fraumeni JF, Jr., et al.: Spectrum of cancer risk among US solid organ transplant recipients. JAMA 2011;306:1891-901.
[7] 7. Na R, Grulich AE, Meagher NS, McCaughan GW, Keogh AM, Vajdic CM: Comparison of de novo cancer incidence in Australian liver, heart and lung transplant recipients. Am J Transplant 2013;13:174-83.
[8] 8. Koshiol J, Pawlish K, Goodman MT, McGlynn KA, Engels EA: Risk of hepatobiliary cancer after solid organ transplant in the United States. Clin Gastroenterol Hepatol 2014;12:1541-9.e3.
[9] 9. Nair N, Gongora E, Mehra MR: Long-term immunosuppression and malignancy in thoracic transplantation: where is the balance? J Heart Lung Transplant 2014;33:461-7.
[10] 10. de Mattos AM, Olyaei AJ, Bennett WM: Nephrotoxicity of immunosuppressive drugs: long-term consequences and challenges for the future. Am J Kidney Dis 2000;35:333-46.
[11] 11. Engels EA: Inflammation in the development of lung cancer: epidemiological evidence. Expert Rev Anticancer Ther 2008;8:605-15.
[12] 12. Grulich AE, van Leeuwen MT, Falster MO, Vajdic CM: Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet 2007;370:59-67.
[13] 13. Guba M, Graeb C, Jauch K-W, Geissler EK: Pro- and anti-cancer effects of immunosuppressive agents used in organ transplantation. Transplantation 2004;77:1777-82.
[14] 14. van Leeuwen MT, Grulich AE, Webster AC, et al.: Immunosuppression and other risk factors for early and late non-Hodgkin lymphoma after kidney transplantation. Blood 2009;114:630-7.
[15] 15. Dell-Olio D, Kelly DA: Immunosuppressants: what's new? Curr Opin Organ Transplant 2010;15:594-600.
[16] 16. Moher D, Liberati A, Tetzlaff J, Altman DG, Group P: Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097-e.
[17] 17. Wells GJhwocpceoa: The Newcastle-Ottawa Scale (NOS) for assessing the quality of non randomised studies in meta-analyses. 2001.
[18] 18. Chalmers ZR, Connelly CF, Fabrizio D, et al.: Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med 2017;9:34-.
[19] 19. DerSimonian R, Laird NJCct: Meta-analysis in clinical trials. 1986;7:177-88.
[20] 20. Ades A, Lu G, Higgins JJMDM: The interpretation of random-effects meta-analysis in decision models. 2005;25:646-54.
[21] 21. Higgins JP, Thompson SG, Deeks JJ, Altman DGJB: Measuring inconsistency in meta-analyses. 2003;327:557-60.
[22] 22. Egger M, Smith GD, Schneider M, Minder CJB: Bias in meta-analysis detected by a simple, graphical test. 1997;315:629-34.
[23] 24. Collett D, Mumford L, Banner NR, Neuberger J, Watson C: Comparison of the incidence of malignancy in recipients of different types of organ: a UK Registry audit. Am J Transplant 2010;10:1889-96.
[24] 25. Ekstrom M, Riise GC, Tanash HA: Risk of cancer after lung transplantation for COPD. International journal of chronic obstructive pulmonary disease 2017;12:2841-7.
[25] 26. Engels EA, Pfeiffer RM, Fraumeni JF, et al.: Spectrum of cancer risk among US solid organ transplant recipients. JAMA 2011;306:1891-901.
[26] 27. Fink AK, Yanik EL, Marshall BC, et al.: Cancer risk among lung transplant recipients with cystic fibrosis. Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society 2017;16:91-7.
[27] 28. Hortlund M, Arroyo Muhr LS, Storm H, Engholm G, Dillner J, Bzhalava D: Cancer risks after solid organ transplantation and after long-term dialysis. Int J Cancer 2017;140:1091-101.
[28] 29. Jäämaa-Holmberg S, Salmela B, Lemström K, Pukkala E, Lommi J: Cancer incidence and mortality after heart transplantation - A population-based national cohort study. Acta Oncol 2019;58:859-63.
[29] 30. Jensen AO, Svaerke C, Farkas D, Pedersen L, Kragballe K, Sorensen HT: Skin cancer risk among solid organ recipients: a nationwide cohort study in Denmark. Acta Derm Venereol 2010;90:474-9.
[30] 31. Jiang Y, Villeneuve PJ, Wielgosz A, Schaubel DE, Fenton SSA, Mao Y: The incidence of cancer in a population-based cohort of Canadian heart transplant recipients. Am J Transplant 2010;10:637-45.
[31] 32. Kellerman L, Neugut A, Burke B, Mancini D: Comparison of the incidence of de novo solid malignancies after heart transplantation to that in the general population. Am J Cardiol 2009;103:562-6.
[32] 33. Krynitz B, Edgren G, Lindelof B, et al.: Risk of skin cancer and other malignancies in kidney, liver, heart and lung transplant recipients 1970 to 2008--a Swedish population-based study. Int J Cancer 2013;132:1429-38.
[33] 34. Krynitz B, Olsson H, Lundh Rozell B, Lindelof B, Edgren G, Smedby KE: Risk of basal cell carcinoma in Swedish organ transplant recipients: a population-based study. Br J Dermatol 2016;174:95-103.
[34] 35. Laprise C, Cahoon EK, Lynch CF, et al.: Risk of lip cancer after solid organ transplantation in the United States. Am J Transplant 2019;19:227-37.
[35] 36. Magruder JT, Crawford TC, Grimm JC, et al.: Risk Factors for De Novo Malignancy Following Lung Transplantation. Am J Transplant 2017;17:227-38.
[36] 37. Morton LM, Gibson TM, Clarke CA, et al.: Risk of myeloid neoplasms after solid organ transplantation. Leukemia 2014;28:2317-23.
[37] 38. Ohman J, Rexius H, Mjornstedt L, et al.: Oral and lip cancer in solid organ transplant patients--a cohort study from a Swedish Transplant Centre. Oral Oncol 2015;51:146-50.
[38] 39. O'Neill JP, Sexton DJ, O'Leary E, et al.: Post-transplant malignancy in solid organ transplant recipients in Ireland, The Irish Transplant Cancer Group. Clin Transplant 2019;33:e13669.
[39] 40. Rizvi SMH, Aagnes B, Holdaas H, et al.: Long-term Change in the Risk of Skin Cancer After Organ Transplantation: A Population-Based Nationwide Cohort Study. JAMA Dermatol 2017;153:1270-7.
[40] 41. Safaeian M, Robbins HA, Berndt SI, Lynch CF, Fraumeni JF, Jr., Engels EA: Risk of Colorectal Cancer After Solid Organ Transplantation in the United States. Am J Transplant 2016;16:960-7.
[41] 42. Serraino D, Piselli P, Busnach G, et al.: Risk of cancer following immunosuppression in organ transplant recipients and in HIV-positive individuals in southern Europe. Eur J Cancer 2007;43:2117-23.
[42] 43. Triplette M, Crothers K, Mahale P, et al.: Risk of lung cancer in lung transplant recipients in the United States. Am J Transplant 2019;19:1478-90.
[43] 44. Tsai H-I, Lee C-W, Kuo C-F, et al.: De novo malignancy in organ transplant recipients in Taiwan: a nationwide cohort population study. Oncotarget 2017;8:36685-95.
[44] 45. Bouvard V, Baan R, Straif K, et al.: A review of human carcinogens--Part B: biological agents. Lancet Oncol 2009;10:321-2.
[45] 46. Mayer V, Ebbesen P: Persistent viral infections in human carcinogenesis. Eur J Cancer Prev 1994;3:5-14.
[46] 47. Buzzeo BD, Heisey DM, Messing EM: Bladder cancer in renal transplant recipients. Urology 1997;50:525-8.
[47] 48. Yan L, Chen P, Chen EZ, Gu A, Jiang ZY: Risk of bladder cancer in renal transplant recipients: a meta-analysis. Br J Cancer 2014;110:1871-7.
[48] 49. Holt CD: Overview of Immunosuppressive Therapy in Solid Organ Transplantation. Anesthesiol Clin 2017;35:365-80.
[49] 50. Sanches MM, Travassos AR, Soares-de-Almeida L: The Relationship Between Immunodepression and the Development of Skin Cancer. Acta Med Port 2017;30:69-72.
[50] 51. Chin-Hong PV, Reid GE, Practice ASTIDCo: Human papillomavirus infection in solid organ transplant recipients: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant 2019;33:e13590-e.
[51] 52. Mittal A, Colegio OR: Skin Cancers in Organ Transplant Recipients. American Journal of Transplantation 2017;17:2509-30.
[52] 53. Brem R, Li F, Karran P: Reactive oxygen species generated by thiopurine/UVA cause irreparable transcription-blocking DNA lesions. Nucleic Acids Res 2009;37:1951-61.
[53] 54. Kelly GE, Meikle W, Sheil AG: Effects of immunosuppressive therapy on the induction of skin tumors by ultraviolet irradiation in hairless mice. Transplantation 1987;44:429-34.
[54] 55. Marzo-Castillejo M, Vela-Vallespín C: Overdiagnosis in cancer. Aten Primaria 2018;50 Suppl 2:51-6.
[55] 56. Stenzinger A, Allen JD, Maas J, et al.: Tumor mutational burden standardization initiatives: Recommendations for consistent tumor mutational burden assessment in clinical samples to guide immunotherapy treatment decisions. Genes Chromosomes Cancer 2019;58:578-88.
[56] 57. Ordóñez-Mena JM, Schöttker B, Mons U, et al.: Quantification of the smoking-associated cancer risk with rate advancement periods: meta-analysis of individual participant data from cohorts of the CHANCES consortium. BMC Med 2016;14:62-.
[57] 58. Freisling H, Arnold M, Soerjomataram I, et al.: Comparison of general obesity and measures of body fat distribution in older adults in relation to cancer risk: meta-analysis of individual participant data of seven prospective cohorts in Europe. Br J Cancer 2017;116:1486-97.
[58] 59. Bagnardi V, Rota M, Botteri E, et al.: Alcohol consumption and site-specific cancer risk: a comprehensive dose-response meta-analysis. Br J Cancer 2015;112:580-93.
[59] 60. Hojo M, Morimoto T, Maluccio M, et al.: Cyclosporine induces cancer progression by a cell-autonomous mechanism. Nature 1999;397:530-4.
There are no comments yet...