Scedosporium apiospermum and lomentospora prolificans in lung transplant patients: A single centre experience over 24 years
Jaideep Vazirani1, Glen P. Westall 1, C. Orla Morrissey 2, Greg I. Snell 1.
1Department of Respiratory Medicine, The Alfred Hospital, Melbourne, Australia; 2Department of Infectious Diseases, The Alfred Hospital, Melbourne, Australia
Introduction: Sceodosporium and Lomentospora (Sceodosporium/Lomentospora) species cause multi resistant and life-threatening respiratory tract infections. Amongst lung transplant recipients, these emerging pathogens now represent the second most common cause of invasive mould infections following Aspergillus.(1,2) The current epidemiology, diagnostic algorithm and management is unknown.
Methods: A retrospective single centre audit of all sputum and bronchoscopy samples positive for Sceodosporium/Lomentospora species in lung transplant patients over a 15-year period (1995-2019). Patients were defined as either being colonised with the pathogen or diagnosed with invasive fungal disease (IFD) as per 2010 the International Society of Heart and Lung Transplant (ISHLT) definitions(3).
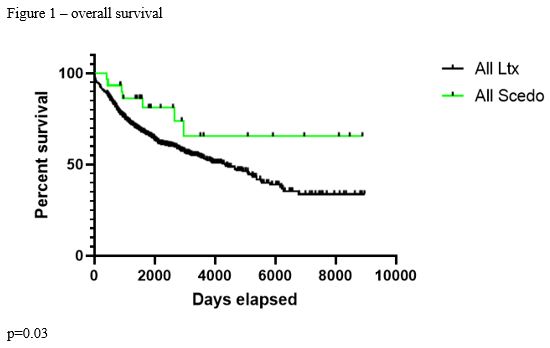
Results: Since 1995, 30 from a total lung transplant cohort of 959 patients (3.13%) cultured Scedosporium/Lomentospora from either sputum sample or bronchoscopic washings. The first isolate was detected in 2014 (n=9), with cases detected yearly thereafter (2015 [n=1]; 2016 [n=8]; 2017 [n=6]; n=2018 [n=6]). Nineteen (63%) Scedosporium/Lomentospora isolation was classified as invasive (IFD), and 27% (n=11) were deemed as colonisers. The median time to first culture from lung transplant was 929 days [95% CI 1089-3168 days]. The majority of patients (n = 19; 63.3%) had received an antifungal agent prior to the first positive culture (posaconazole [n=15; 79%], voriconazole [n=2;11%], posaconazole + terbinadine [n=1; 5%], terbinafine [n=1; 5%]). The most common antifungal agent used for treatment was posaconazole (n=16) followed by terbinafine (n=9), and multiple sequential agents were used in 8 (26.7%). Median duration of therapy was 364 days [95% CI 264-748 days]. Median FEV1 increased by 400mls following six months treatment of Scedosporium/Lomentospora.
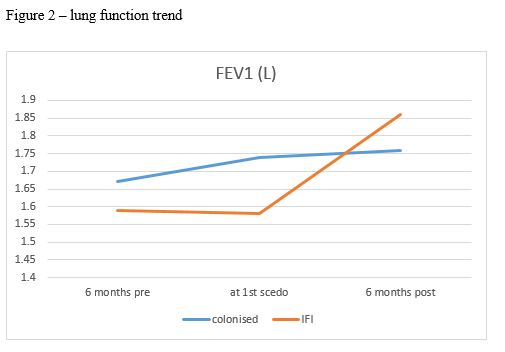
Overall survival amongst patients with Scedosporium/Lomentospora detected from respiratory sampling superior compared to the remainder of the LTx cohort (65% vs 34% p=0.04)
Discussion: Our experience is in keeping with previous smaller studies, however this sample size and duration of follow up for Scedosporum/Lomentospora has not previously been reported. Our single centre 24-year experience suggests that the incidence of Scedosporium/Lomentospora in respiratory samples of lung transplant recipients is emerging. Most patients with positive Scedosporum/Lomentospora on sputum/bronchoscopic washings have had prior exposure to anti-fungal agents.
Conclusion: Treatment of Scedosporium/Lomentospora infection results in improved lung function, and in good overall survival rates. Further surveillance is required to fully elucidate the impact of these organisms on lung transplant recipients.
[1] Pappas PG, Alexander BD, Andes DR, Hadley S, Kauffman CA, Freifeld A, et al. Invasive Fungal Infections among Organ Transplant Recipients: Results of the Transplant‐Associated Infection Surveillance Network (TRANSNET). CLIN INFECT DIS. 2010 Apr 15;50(8):1101–11
[2] Rammaert B, Puyade M, Cornely OA, Seidel D, Grossi P, Husain S, et al. Perspectives on Scedosporium species and Lomentospora prolificans in lung transplantation: Results of an international practice survey from ESCMID fungal infection study group and study group for infections in compromised hosts, and European Confederation of Medical Mycology. Transpl Infect Dis [Internet]. 2019 Oct [cited 2020 Feb 24];21(5). Available from: https://onlinelibrary.wiley.com/doi/abs/10.1111/tid.13141
[3] Husain S, Mooney ML, Danziger-Isakov L, Mattner F, Singh N, Avery R, et al. A 2010 working formulation for the standardization of definitions of infections in cardiothoracic transplant recipients. The Journal of Heart and Lung Transplantation. 2011 Apr;30(4):361–74.
There are no comments yet...