Successful long-term TMA- and rejection-free survival of a kidney xenograft with triple xenoantigen knockout plus insertion of multiple human transgenes
David Ma1, Takayuki Hirose1, Ivy Rosales1, Hajime Sasaki1, Robert Colvin1, Jim Markmann1,2, Wenning Qin2, Yinan Kan2, Jacob Layer2, Michele Youd2, William Westlin2, Luhan Yang2, Tatsuo Kawai1.
1Center for Transplant Sciences, Massachusetts General Hospital, Boston, MA, United States; 2eGenesis Inc., Cambridge, MA, United States
Usage of porcine organs is a potential solution to the shortage of organs for transplantation. However it continues to be hindered by multiple immunologic barriers. Recent advances in genome engineering technology allow for the generation of porcine organ donors with multiplex gene edits. Here we evaluated survival of kidneys from triple xenoantigen knockout pigs that contain multiple human transgenes (Pig 2.0) in a life-sustaining non-human primate renal xenotransplant model.
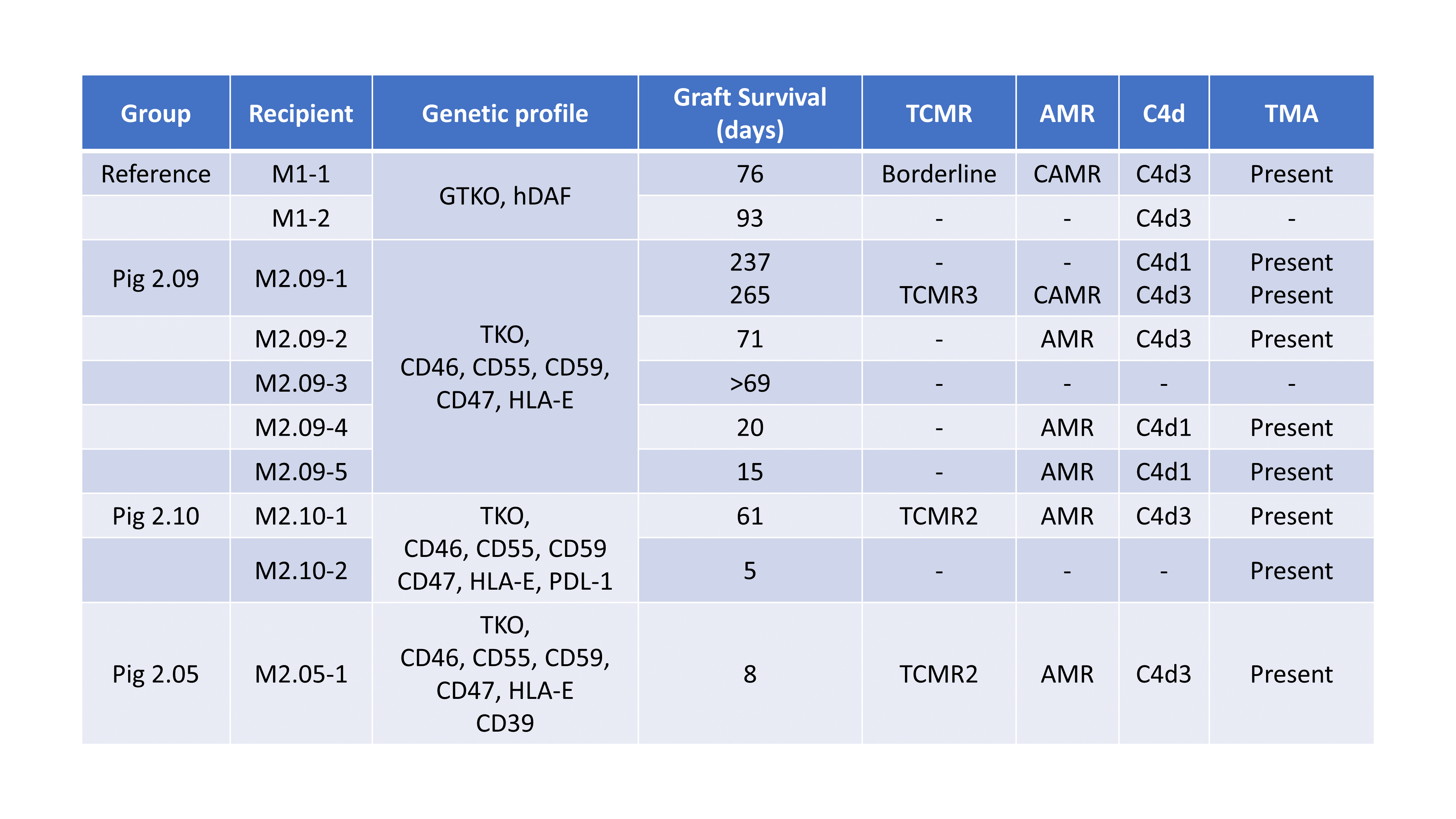
Cynomolgus macaques received kidneys from either GTKO.hCD55 pigs (n=2; reference group) or Pig 2.0 (n=8) followed by bilateral native nephrectomy. All Pig 2.0 donors were triple antigen knockout and expressed human complement, inflammation, and immune regulatory transgenes at different levels in three variations (Pig 2.5, 2.9, 2.10). Recipients were treated with anti-rhesus ATG and anti-CD20 mAb as induction, followed by weekly anti-CD154 (hu5C8), daily mycophenolate mofetil (MMF) and, in the reference and 2.9 groups, methylprednisolone and short-term sirolimus. Animals were followed clinically with lab tests, serial ultrasound and protocol biopsies.
Regardless of gene modifications, graft survival clustered into three patterns – rapid rejection within three weeks (n=4), delayed rejection at two months (n=3), and longer-term surviving where the animals were euthanized for non-rejection related complications (n=2). One animal remains on study at day 69 without evidence of rejection on his first protocol biopsy. The longest survivor (Pig 2.9) had no evidence of rejection on biopsy at day 237. MMF was discontinued on day 245 due to parvovirus-induced anemia. This intractable anemia led to sacrifice of the animal on day 265. Autopsy revealed TCMR, chronic AMR and TMA. Overall, graft rejection was associated with thrombotic microangiopathy (TMA), chronic antibody-mediated rejection (AMR) and T-cell Mediated Rejection (TCMR). Recipients were not screened for preformed anti-pig antibodies. Retrospective analysis showed longer-term survivors had lower IgM and IgG (MFI<500 and <2000, respectively) pre-transplant antibody levels against donor endothelial cells than delayed (MFI >3000 and >8500) or rapid rejection groups (MFI >4500 and >8500). Two animals in the delayed rejection group revealed a small number of CD20+ B cells in autopsy samples on days 61 and 71, respectively.
Genetic modification with depletion of xenoantigens and addition of human transgenes allowed kidney graft survival up to 265 days in the Pig 2.0 group. Lower pre-transplant donor specific antibody titers were generally associated with better post-transplant outcomes.