Podocyte-specific urinary extracellular vesicles: A novel biomarker for recurrent primary focal segmental glomerulosclerosis after kidney transplantation
Hien Lau1, Shiri Li1, Frank Zaldivar2, Akihiro Shimomura1, Uttam G. Reddy3, Ekamol Tantisattamo3, Donald Dafoe1, Hirohito Ichii1.
1Department of Surgery, University of California, Irvine, Irvine, CA, United States; 2Pediatric Exercise and Genomics Research Center, University of California, Irvine, Irvine, CA, United States; 3Division of Nephrology and Hypertension, Department of Medicine, University of California, Irvine, Irvine, CA, United States
Introduction: Recurrent primary focal segmental glomerulosclerosis (FSGS) after kidney transplantation (KT) involving podocyte injury and depletion is a common and potential cause of renal allograft loss, which has been associated with an increased mortality rate after return to dialysis. Renal allograft biopsy is the gold standard to diagnose FSGS recurrence; however, it is costly and invasive. Non-invasive methods that can detect podocyte injury may provide early diagnosis and guide prompt management to enhance graft survival. Urinary extracellular vesicles (UEVs) secreted by various types of renal cells contain cell-specific molecular cargos that may serve as an attractive source of diagnostic biomarkers for kidney diseases. This study aims to measure the level of podocyte-specific UEVs in renal transplant recipients (RTRs) with recurrent primary FSGS and correlate this level to renal allograft function.
Materials and Methods: UEVs from urine samples of healthy control RTRs (CTL, n=5), RTRs without FSGS recurrence (NR-FSGS, n=4), and RTRs with FSGS recurrence (FSGS, n=4) were isolated within 24 hours. FSGS recurrence was confirmed by severe proteinuria and renal allograft biopsy. Isolated UEVs were stained with Annexin V, CD63 antibody, and podocalyxin antibody. Stained UEVs were analyzed on a flow cytometer and expressed as both percentage and total number of UEVs normalized to urine creatinine.
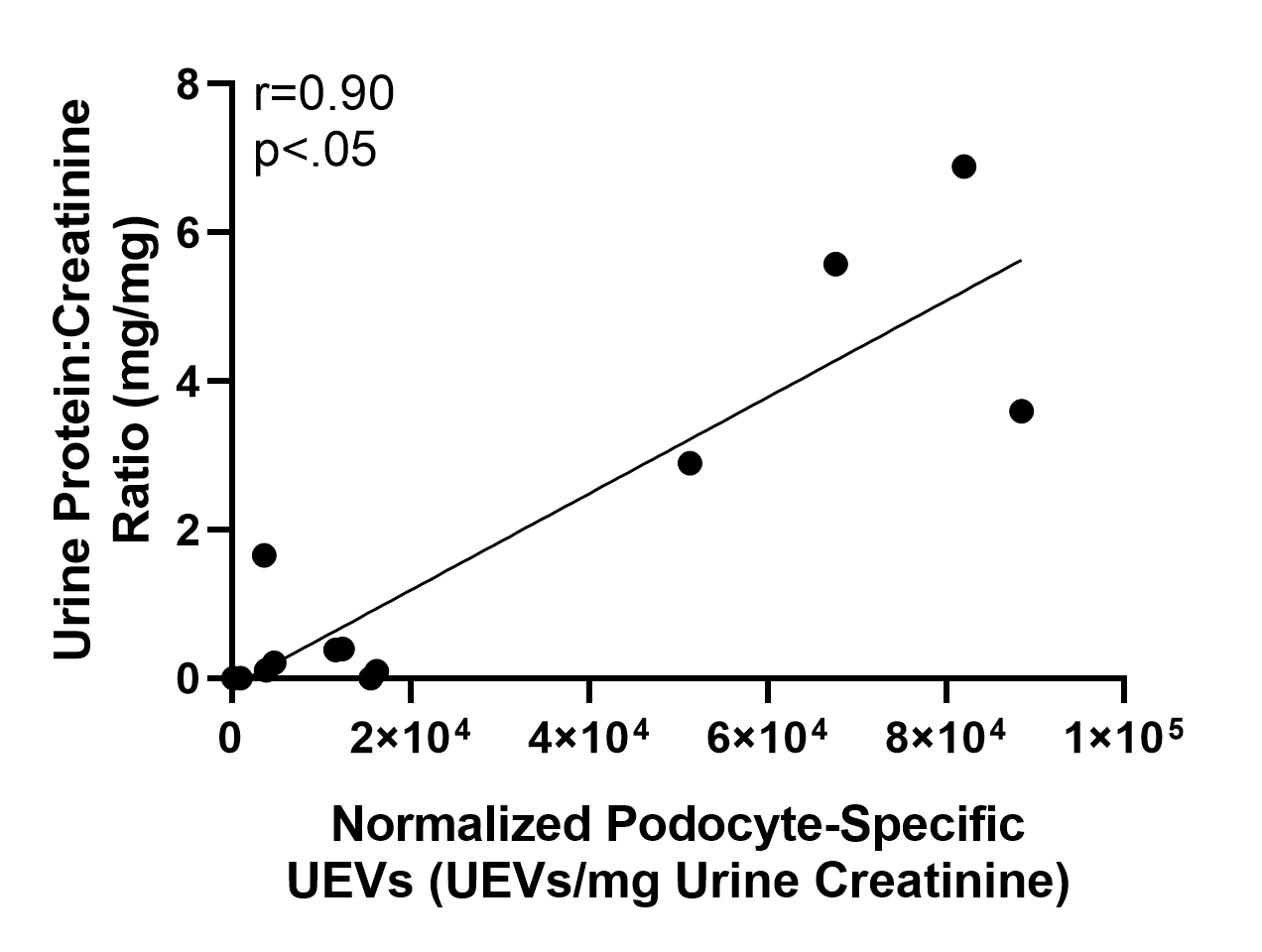
Results and Discussion: The percentage of Annexin V-positive UEVs (CTL=50.87±11.22%, NR-FSGS=46.30±13.29%, FSGS=67.11±5.69%, p=NS) and CD63-positive UEVs (CTL=82.88±3.98%, NR-FSGS=77.65±6.53%, FSGS=83.10±3.71%, p=NS) were similar among the three groups. FSGS group had a significantly higher number of normalized Annexin V-positive UEVs compared to CTL and NR-FSGS groups (CTL=6.90±2.16x104 UEVs/mg, NR-FSGS=10.27±2.73x104 UEVs/mg, FSGS=27.39±5.26x104 UEVs/mg, p<.05). The percentage (CTL=4.87±1.93%, NR-FSGS=6.90±3.73%, FSGS=30.80±6.52%, p<.05) and normalized number of podocyte-specific UEVs (CTL=5.55±2.61x103 UEVs/mg, NR-FSGS=10.27±3.28x103 UEVs/mg, FSGS=72.36±8.25x103 UEVs/mg, p<.05) were significantly elevated in FSGS group compared to remaining groups. The normalized number of podocyte-specific UEVs was positively correlated to SCr (r=0.58, p<.05) and UPCR (r=0.90, p<.05).
Conclusion: A significantly high level of podocyte-specific UEVs and its strongly positive correlation with SCr and proteinuria as clinical evidences of renal allograft injury occurred in RTRs with recurrent primary FSGS, but not in healthy control RTRs or RTRs without recurrent FSGS. Our findings suggest that cell-specific UEVs may serve as a novel non-invasive biomarker for podocyte-specific injuries in RTRs with recurrent primary FSGS.
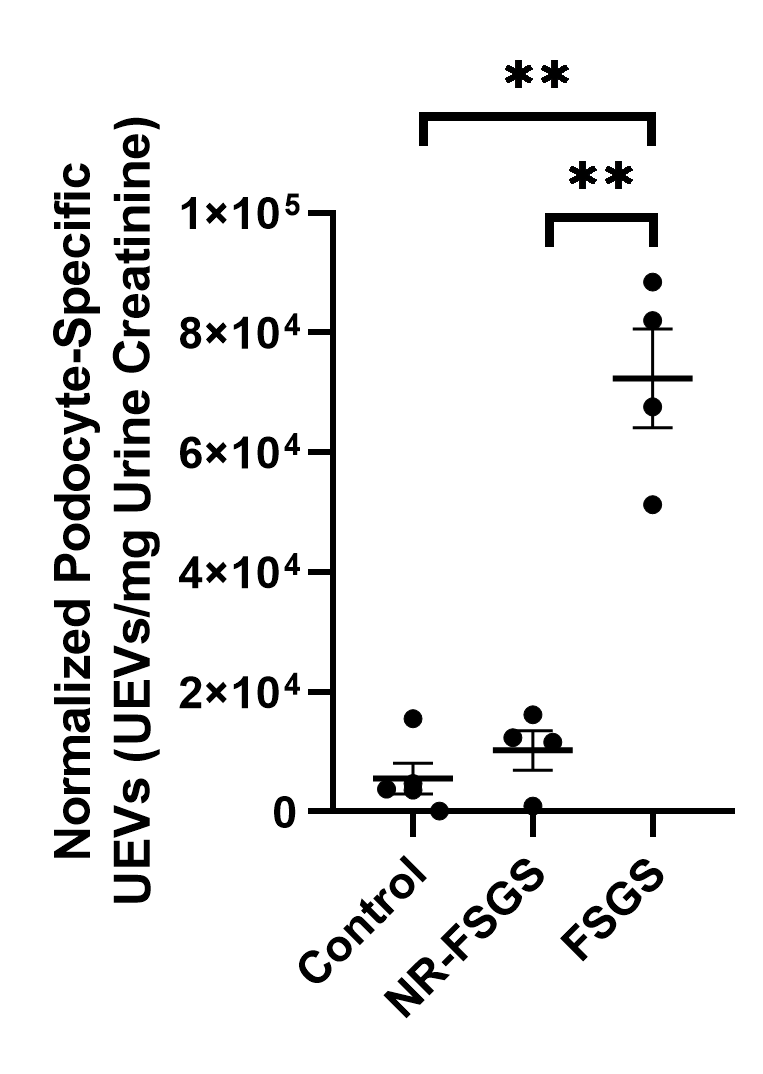
There are no comments yet...