The impact of oxygen free radical scavenger on acute rejection in a histocompatible miniature swine model
Philipp Tratnig-Frankl1,2, Alec R. Andrews1, Corentin B. Taveau1,2,3, Alexandre G. Lellouch1,2,3, Michael M. Jonczyk1,2,4, Marion P. Goutard1,2,3, Elise L. Lupon1,2,5, Mark A. Randolph1,2, Michael L. Morrison6, Mark B. Roth6, Curtis L. Cetrulo Jr.1,2.
1Division of Plastic Surgery, Massachusetts General Hospital, Harvard Medical School, Boston, MA, United States; 2Division of Plastic Surgery , Shriners Hospitals for Children, Boston, MA, United States; 3Service de Chirurgie Plastique, Hôpital Européen Georges Pompidou, Assistance Publique - Hôpitaux de Paris (APHP), Paris, France; 4Sackler Graduate School CTSI Program, Tufts University, Boston, MA, United States; 5Service de Chirurgie Plastique, Rangueil Hospital, University Toulouse III Paul Sabatier, Toulouse, France; 6Fred Hutchinson Cancer Research Center, Seattle, WA, United States
Introduction: Vascularized Composite Allotransplantations (VCAs), like solid organ transplants, are very susceptible to ischemia-reperfusion injury (IRI). IRI leads to tissue hypoxia and the formation of oxygen free radicals which triggers T-cell accumulation and complement activation, causing increased antigenicity of the transplanted tissue.
In this study we demonstrate the effect of two oxygen free radical scavengers on acute rejection in a histocompatible miniature swine model.
Materials and Methods: To conduct this study, we used our well-established myocutaneous flap model in a major histocompatibility complex (MHC)-defined miniature swine. MHC class I and II antigens were matched between donors and recipients. Myocutaneous allografts consisted of an isolated skin pad with the underlying gracilis muscle. All flaps were treated with similar concentrations of either Saline (9mg/ml, n=2), Sodium Iodide (10mg/ml, n=3) or Hydrogen Sulfide (H2S) (11.6mg/ml). Flaps were subjected to an intermediate ischemia time of 3 hours at 4°C immediately after harvest. End of ischemia time was marked by unclamping the anastomosis. Daily blood draws were performed on postoperative day (POD) 0 to 7, 14 and end of study. Biopsies for histological analysis were collected on POD 4, 10, 14 and end of study. Rejection was assessed by an independent pathologist following the Banff criteria (Grade 0-IV) for skin containing VCA.
Results: On POD 4, flaps treated with H2S showed Grade 0 of rejection according to the Banff classification, compared to Grade I in the Sodium Iodide group and Grade II in the Saline. There was no significant difference in grade of rejection between these three groups on or after POD 10.
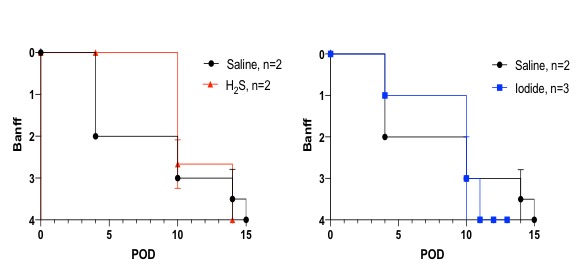
Discussion: Treatment of ischemic myocutaneous flaps with oxygen free radical scavengers delayed acute rejection when compared to saline treated controls. Currently, standard immunosuppression regimens and techniques for graft preservation of VCA do not enable preconditioning of the donor or ischemic-free storage. Therefore, IRI is unavoidable in VCA so far. Diminishing the impact of IRI could reduce local secretion of pro-inflammatory cytokines and sequester recruitment of alloreactive immune cells to the transplant, delaying acute rejection episodes during the immediate post-operative period.
Conclusion: With this study we demonstrate the impact of oxygen free radical scavengers on IRI in an immunocompetent and genetically well-defined large animal model. Treating myocutaneous flaps accordingly abrogates early rejection histopathologically at POD 4. Further experiments are ongoing to titrate the impact of these scavengers across later timepoints.
[1] Eltzschig HK, Eckle T. Ischemia and reperfusion--from mechanism to translation. Nat Med. 2011;17(11):1391-401.
[2] Cendales LC, Kanitakis J, Schneeberger S, Burns C, Ruiz P, Landin L, et al. The Banff 2007 working classification of skin-containing composite tissue allograft pathology. Am J Transplant. 2008;8(7):1396-400.
[3] Leto Barone AA, Leonard DA, Torabi R, Mallard C, Glor T, Scalea JR, Randolph MA, Sachs DH, Cetrulo CL Jr. The gracilis myocutaneous free flap in swine: an advantageous preclinical model for vascularized composite allograft transplantation research. Microsurgery. 2013;33(1):51-5.
There are no comments yet...