
Pre-kidney transplant latent tuberculosis infection screening and posttransplant active tuberculosis
Gisele Meinerz1,2, Cynthia da Silva1, Damaris Dorsdt4, Julia Adames4, Julio Andrade4, Pedro Ventura4, Alexandre Monteiro3, Alessandro Pasqualotto3, Valter Garcia1, Elizete Keitel1,2.
1Nephrology and Kidney and Transplant Department, ISCMPA, Porto Alegre, Brazil; 2Post Graduation Program in Pathology, UFCSPA, Porto Alegre, Brazil; 3ISCMPA, Porto Alegre, Brazil; 4UFCSPA, Porto Alegre, Brazil
Background: Tuberculosis (TB) is an important infectious complication after kidney transplantation (KT). Latent tuberculosis infection (LTI) screening and treatment is fundamental to prevent post-transplant active disease.
Methods: We described a cohort of adult KT recipients from April 4th, 2014 to October 31st, 2018, with follow-up until October 31st, 2019, regarding pre-transplant LTI screening and posttransplant active tuberculosis.
Results: There were 986 KT performed in the study period. There were 189 KT with positive LTI screening, 488 with negative and 309 with incomplete screening.
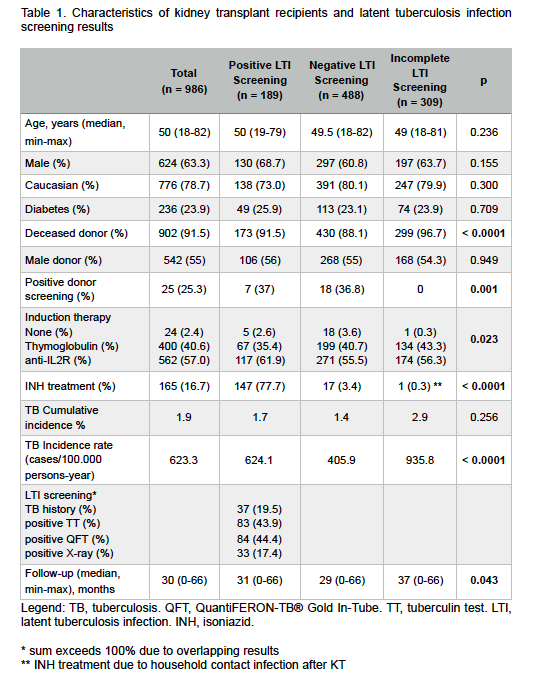
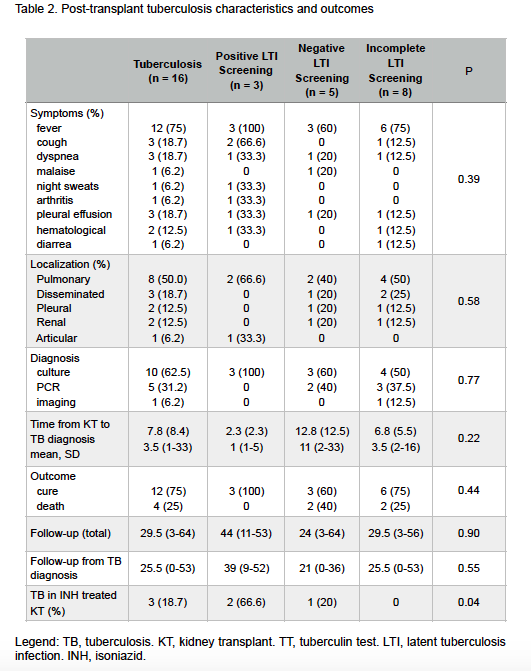
Most KT recipients were male (63.3%), Caucasian (78.7%), with deceased donors (91.5%). Median age was 50yo. LTI treatment was provided to 79% of patients with indication, and 1.8% of them developed active TB. There were 16 cases of active tuberculosis, 50% pulmonary.
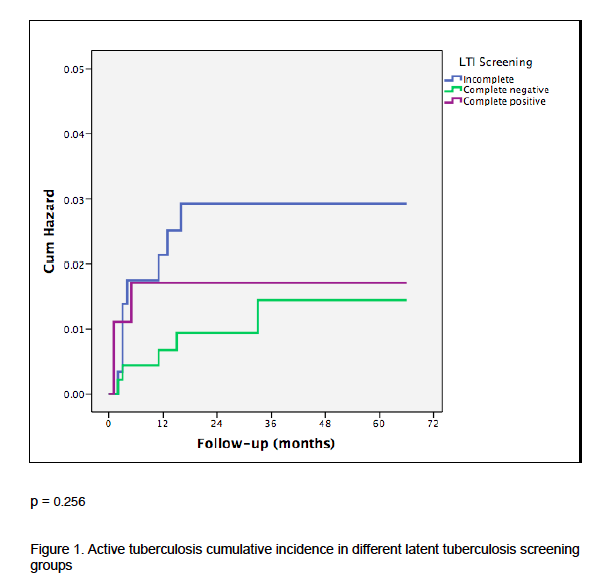
Cumulative 5-year TB incidence in positive LTI screening patients was 1.7%; in patients with negative screening was 1.4%, and in those with incomplete screening was 2.9% (p = 0.25).
TB incidence rate in positive LTI screening patients was 624.1 cases per 100.000 patients-year; in negative screening was 405.9 cases per 100.000 patients-year; and in incomplete screening was 935.8 cases per 100.000 patients-year (p < 0.0001). TB patients had inferior although non-significant five-year graft survival (62.5% vs. 77.4%, p = 0.07). Twenty-five percent of TB patients died.
Conclusion: One third of our patients had incomplete LTI screening, and this group had a significantly higher TB incidence rate. A considerable proportion of patients with positive LTI screening completed INH treatment. Only 1.3% of KT that received INH treatment developed TB, in a short period after transplantation. There was a high incidence of post-transplant TB in this cohort and high mortality.
Fapergs - Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul.
[1] Aguado JM, Torre-Cisneros J, Fortun J, Benito N, Meije Y, Doblas A, et al. Tuberculosis in solid-organ transplant recipients: consensus statement of the group for the study of infection in transplant recipients (GESITRA) of the Spanish Society of Infectious Diseases and Clinical Microbiology. Clin Infect Dis. 2009;48(9):1276-84.
[2] Abad CLR, Razonable RR. Mycobacterium tuberculosis after solid organ transplantation: A review of more than 2000 cases. Clin Transplant. 2018:e13259.
[3] Subramanian AK, Theodoropoulos NM, Infectious Diseases Community of Practice of the American Society of T. Mycobacterium tuberculosis infections in solid organ transplantation: Guidelines from the infectious diseases community of practice of the American Society of Transplantation. Clin Transplant. 2019;33(9):e13513.
[4] WHO. Global tuberculosis report 2018. Geneva: World Health Organization; 2018. Licence: CCBY-NC-SA3.0IGO. 2019.
[5] Currie AC, Knight SR, Morris PJ. Tuberculosis in renal transplant recipients: the evidence for prophylaxis. Transplantation. 2010;90(7):695-704.
[6] Adamu B, Abdu A, Abba AA, Borodo MM, Tleyjeh IM. Antibiotic prophylaxis for preventing post solid organ transplant tuberculosis (Review). Cochrane Database of Systematic Reviews. 2014(3).
[7] Abad CLR, Deziel PJ, Razonable RR. Treatment of latent TB Infection and the risk of tuberculosis after solid organ transplantation: Comprehensive review. Transpl Infect Dis. 2019;00:e13178.
There are no comments yet...